Macrocycles via Pnictogen-directed Self-assembly
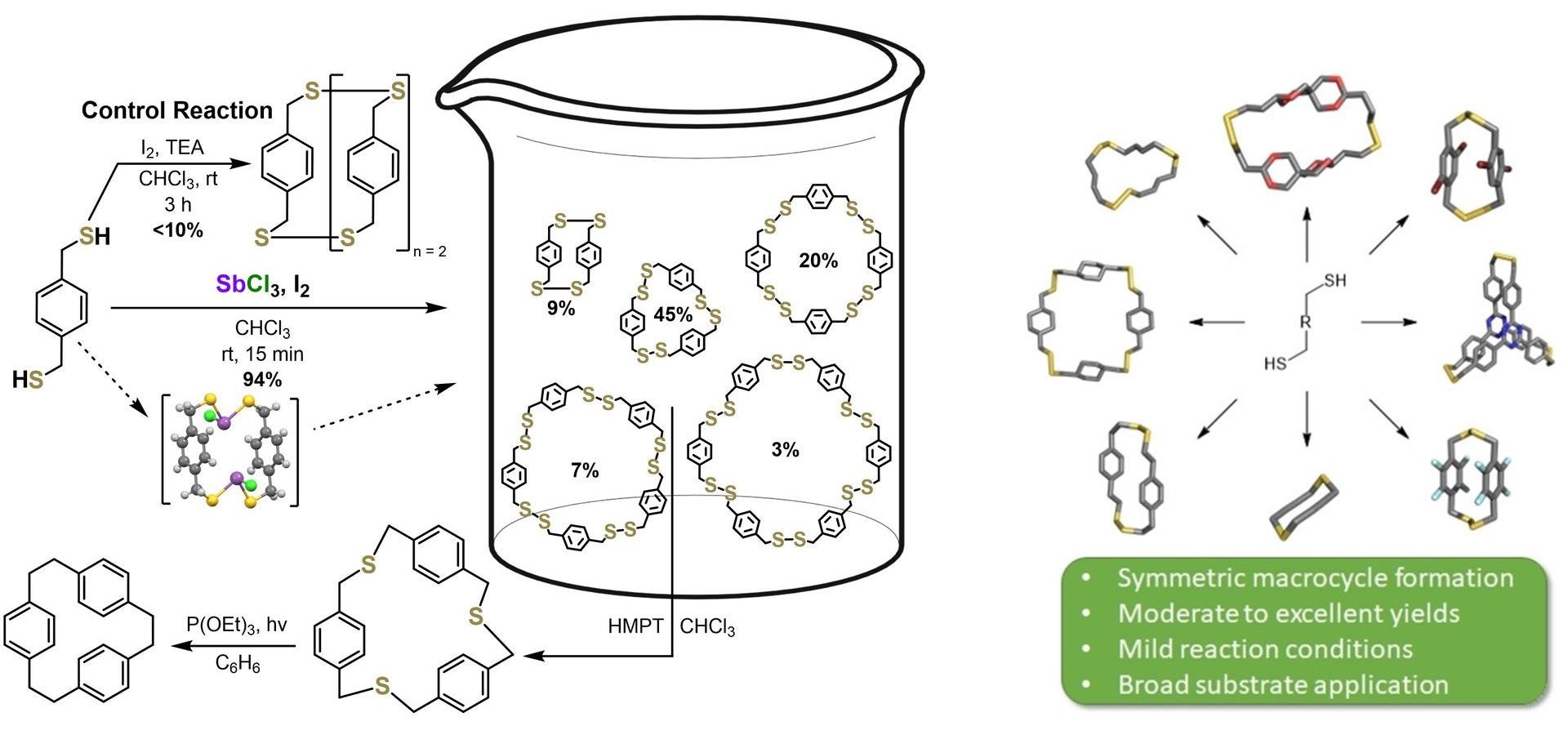
Macrocycles are cyclic molecules that often feature large, functional interior cavities among other unique structural properties. These molecules have many applications including chemical sensing, environmental remediation, supramolecular polymers, and are observed within biological systems. Synthesizing macrocyclic molecules can often be challenging due to high synthetic complexity as well as the kinetically unfavorable ring closure step which often results in undesired oligomers and low yields of macrocycle. Here, we are focused on utilizing pnictogen-directed self-assembly to form novel and otherwise difficult to synthesize macrocyclic systems in high yields. This technique utilizes thiol-bearing monomers to form disulfide macrocycles in high yields and under mild conditions. These disulfide macrocycles can then be separated by size exclusion chromatography, sulfur extruded to thioethers, and further sulfur extruded to the corresponding hydrocarbon. Current work is focused on expanding this self-assembly technique to functionalized macrocycles, and fundamental investigations into the effects of the self-assembly conditions on macrocycle formation.
Key papers:
- Zocchi, L. J.; Davis, W. A.; Zakharov, L. N.; Johnson, D. W. Expanding Pnictogen-Assisted Self-Assembly of Disulfide Macrocycles to Include Heteroarenes. J. Porphyr. Phthalocyanines. 2023, 27, 1394–1397.
- Shear, T. A., Mayhugh, J. T., Zocchi, L. J., Demachkie, I. S., Trubenstein, H. J., Zakharov, Z. N., Johnson, D. W., Expanding the Scope of Pnictogen-Assisted Cylcophane Self-Assembly. Eur. J. Org. Chem. 2022, e202200056.
- Shear, T. S., Johnson, D. W.; Main Group Supramolecular Chemistry Led to Surprising New Directions in the Self-Assembly of Organic Macrocycles, Cages, and Cyclophanes. Synlett. 2021. 32, 1702-1710.
- Collins, M. S.; Carnes, M. E.; Nell, B. P.; Zakharov, L. N.; Johnson, D. W. A Facile Route to Old and New Cyclophanes Via Self-Assembly and Capture Nature Comm. 2016, 7, 11052.
- Collins, M.S.; Phan, M.-N.; Zakharov, L.N.; Johnson, D.W. Coupling Metaloid-Directed Self-Assembly and Dynamic Covalent Systems as a Route to Large Organic Cages and Cyclophanes.
Inorg. Chem. 2018,
57,
3486-3496.